Bias in Clinical Trials
Completion requirements
View
1. Where Bias Can Be Introduced
1.1. During Participant Recruitment
Selection bias
Selection bias can occur at different levels. Some examples are given below:
- If the participant eligibility criteria used in a trial are so stringent that only a small percentage of the intended patient population qualifies. For example, those with better physical condition who would be expected to be able to manage toxic treatments.
- At treatment assignment, the eligibility criteria used to recruit and enrol participants into the treatment arms may be applied differently. This may result in some characteristics being over-expressed in one arm compared to the other. For example, if participants were selected differently according to their age or health status. The treatment outcomes may be stronger in the arm where the participants are younger and in a better shape. Therefore, any difference in outcome between the two treatment arms can no longer be attributed only to the received treatment.
- The use of random participant allocation techniques to two or more treatment arms is key to avoiding this type of bias. Randomisation aims to ensure that the treatment arms are comparable both in terms of known and unknown prognostic factors especially over a large number of participants. Well-performed randomisation will allow the researcher to consider the observed treatment effects (response rate, survival, etc.) to be related to the treatment itself and not due to other factors. The treatment allocation should be independent from researchers’ beliefs and expectations. The optimal way is by using a central randomisation algorithm to distribute characteristics across the treatment arms.
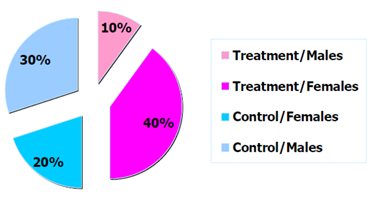
Figure 1: Example distribution of trial participants by arm and sex.
Note that in a trial of limited sample size , there is no guarantee that the randomisation will effectively prevent imbalances in important prognostic factors (age or disease status). In this case, the randomisation can be stratified according to a number of known important prognostic factors. The trial population will be split in groups according to these stratification factors and the randomisation will ensure that the balance between the treatment arms within these strata is maintained.
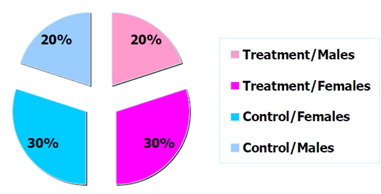
Figure 2: Example distribution of trial participants by arm and sex.