5. Benefit-risk evaluation at the time of approval
Benefit-risk evaluation at the time of approval
The final and most important step in the evaluation of a new medicine is the benefit-risk evaluation. Benefits and risks are considered after assurance that quality, safety and efficacy have been sufficiently documented. The benefit-risk evaluation is a complex process. The present legislation and guidance to industry is not very concrete when it comes to benefit-risk evaluation compared to the other areas.
The assessment report on new medicines has a specific section on the evaluation of the benefit-risk ratio. This is publicly available for individual medicines in the ‘European Public Assessment Report (EPAR) summary for the public’. An EPAR is published for every human or veterinary medicine application that has been granted or refused a marketing authorisation. An EPAR provides public information on a medicine, including how it was assessed by EMA. The EPAR is referred to in Article 13(3) of Regulation (EC) No 726/2004, which requires EMA to publish a public assessment report for each centrally authorised medicine together with a public-friendly overview.
An EPAR is not a single document but an information resource containing several components, including a core set of regulatory documents. The individual components can be viewed online, downloaded and/or printed out. Information handled during the scientific assessment which is considered confidential is removed before an EPAR is published. EPARs are updated periodically to reflect the latest regulatory information on medicines. If the original terms and conditions of a marketing authorisation are modified, the EPAR is updated to reflect such changes with an appropriate level of detail.
EPARs are displayed on the EMA website using four different sections containing different components of the EPAR. The below table provides an overview:
|
Section |
Type of information |
|
Overview |
Public-friendly overview in question-and-answer format. |
|
Authorisation details |
Key details about the product and the marketing authorisation holder. |
|
Package leaflet and summary of product characteristics; labelling; list of all authorised presentations; pharmacotherapeutic group; therapeutic indications. |
|
|
Assessment history |
Public assessment report for the initial authorisation; public assessment report(s) for any variation concerning major changes to the marketing authorisation; orphan maintenance assessment report or withdrawal assessment report (as of 17 January 2018); tabulated overview of procedural steps taken before and after authorisation. |
EPARs can be found on the EMA website (Figure 1). The assessment is written in general terms and the following aspects are summarised:
- Benefits - the positive effects of the medicine on the disease.
- Risks - adverse reactions caused by the medicine.
For an approved medicine, the conclusion will typically be a statement that the benefits outweigh the risks.
Historically there was much less information available, it has been said that the benefit-risk assessment was a matter of personal judgement on the part of the assessors and committees rather than an evidence-based judgement.
It was recognised that a more structured approach was needed for the highly complex benefit-risk assessment. In 2008, the EMA started a benefit-risk methodology project to explore better tools for the process. They created a benefit-risk working party. The working party has analysed a series of more structured tools already available to assess benefit-risk. In 2012, the EMA started a process of training assessors in using structured tools in their assessment work.
The assessment of the benefit-risk balance is a combination of quantitative and qualitative methodologies. Comparative assessment tables, known as Effect Tables, are part of the European Public Assessment Reports (EPARs). Click here for an example.
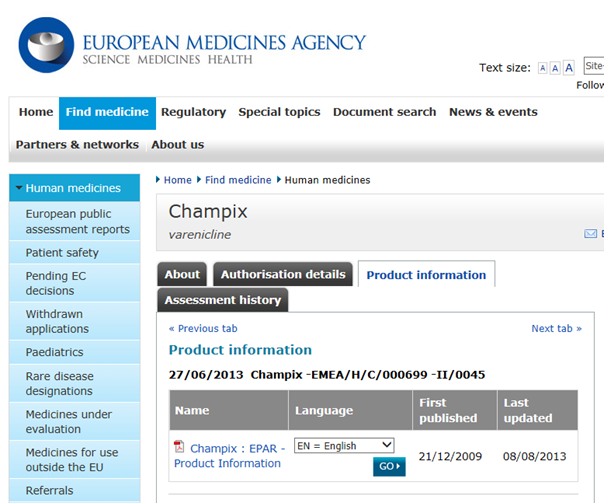
Figure 1: European Public Assessment Report (EPAR) summaries can be found on the EMA website.