Vaccines
| Site: | EUPATI Open Classroom |
| Course: | Types of Medicines and Their Mode of Action and Use |
| Book: | Vaccines |
| Printed by: | Guest user |
| Date: | Friday, 5 June 2026, 2:10 AM |
1. General Features
(This section is organised in the form of a book, please follow the blue arrows to navigate through the book or by following the navigation panel on the right side of the page.)
In contrast to chemical medicines, vaccines usually exert their effects at or near the site of their introduction into the body. They do not need to be distributed through the bloodstream. Individuals often experience redness , tenderness or swelling at the site of the injection, and it is also common to have a slightly raised temperature following vaccination . Serious side effects are very rare. The risks associated with contracting the disease the vaccine prevents are considered to be greater than the risks associated with giving the vaccine. The same dose often works for most people, whatever their age, because of the way vaccines work.2. Mode of Action
To understand how vaccines work, we need to be aware of how the body fights infections. Diseases such as influenza (often called ‘flu’), which are caused by microorganisms such as viruses and bacteria, stimulate a response from the body’s immune system. White blood cells are activated to destroy the invading microorganisms and any infected cells. The immune system can, however, take over a week to mount a response when it is first exposed to a particular type of disease. Some diseases progress rapidly and may harm or even kill patients before the immune system has time to respond. Fortunately, once white blood cells have encountered a particular disease, they develop an immunological memory. This means that if someone is infected with the same disease again, the immune system will quickly recognise the responsible microorganism and respond, so that it is destroyed before symptoms develop.Microorganisms have unique patterns of molecules on their surface known as antigens, which are like fingerprints. The white blood cells in the body specifically recognise these antigens as ‘foreign’. This results in the activation of white blood cells to produce antibodies. Antibodies are specialised proteins that bind to the antigens on the surface of a microorganism, causing it to be attacked and destroyed by the rest of the body’s immune system.
Vaccines work by mimicking a particular disease. During vaccination, a modified form of a disease-causing microorganism or toxin, which does not have the capacity to grow or cause symptoms, is introduced into the body (Figure 1). The vaccine does, however, display the antigens that characterise a particular virus or bacterium. The immune system therefore responds in the same way that it would to the natural form of the disease-causing microorganism or toxin. It mounts a response and learns to recognise the antigens from the microorganisms within the vaccine, so that if the body is later infected with the active disease-causing microorganism it will identify it as being harmful and will act rapidly to destroy it.
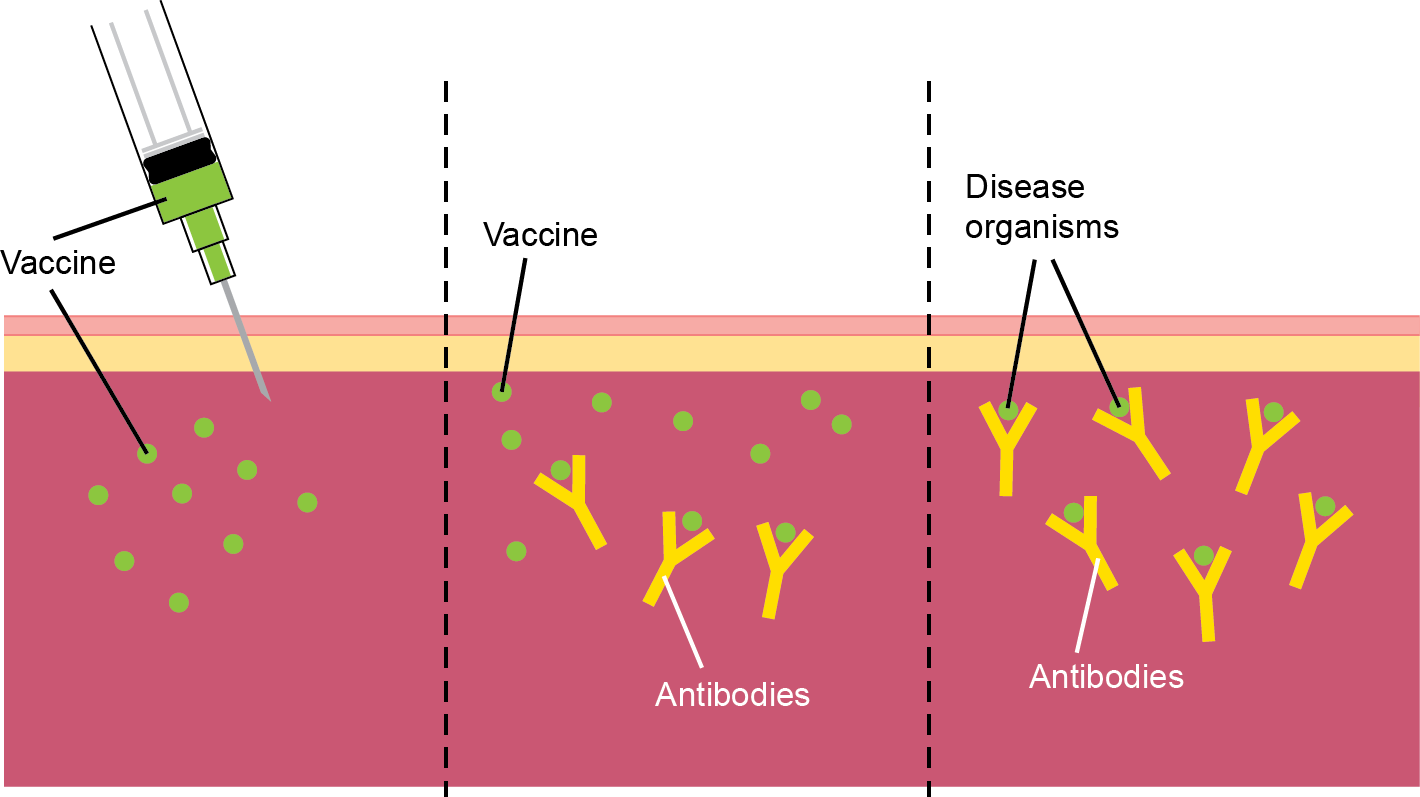
Figure 1. Vaccine immunity. During vaccination, a vaccine with modified forms of viruses or bacteria is injected into the body (left). The vaccine stimulates the immune system to produce antibodies against the microorganism (centre). The immune system learns to recognise the microorganism, so that if the body is later infected with the live disease it will produce antibodies to attach to the microorganisms and stop the infection (right).
Most vaccines are used to prevent disease; however, some can be used to treat disease (known as therapeutic vaccines). For example, there is some evidence that the immune system can be harnessed to kill cancer cells. Tumour cells are derived from the body’s own cells, so under most circumstances the immune system does not see cancer cells as foreign and does not attack the tumour. Cancer vaccines aim to re-educate the immune cells so that they recognise tumour-specific molecules (called tumour antigens) in the same way that they recognise viruses or bacteria. One method under investigation is the use of tumour cells or tumour antigens in a vaccine, combined with a substance that enhances the immune response (an adjuvant) to stimulate the immune system and encourage it to see the tumour as foreign.
3. Production Process
The first step in vaccine development is to grow a modified version of the microorganism that causes the disease. It is modified so that the immune system can still recognize it, but so that it no longer causes harm. There are three main types of vaccine: live-attenuated, inactivated and sub-unit.Live-attenuated vaccines are made from live microorganisms that have been modified to weaken them so that they do not cause disease. These types of vaccine are very good activators of the immune response because they are very similar to the natural infection.
They may not, however, be appropriate for people with a weakened immune system (e.g. individuals with human immunodeficiency virus (HIV) or those who
are receiving chemotherapy). Another disadvantage is that the vaccines need to be kept cool and therefore may not be suitable if no refrigeration is available.
Alternatively, vaccines can contain viruses or bacteria that have been inactivated using chemicals, radiation or heat. These are known as inactivated vaccines. These are more stable than live-attenuated
vaccines and do not require refrigeration, so can be distributed more easily; however, they are not quite as effective at stimulating the immune system as live-attenuated vaccines, so repeat vaccinations (boosters) may be required after a few years.
Sub-unit vaccines contain selected antigens from a microorganism that are able to provoke an immune response. A fourth, but less common, type of vaccine is made from the toxins produced by microorganisms, rather than parts of the microorganism itself.
As of the start of 2014, a number of new approaches to vaccination are being researched. These included the use of DNA vaccines. Individuals are vaccinated
with DNA containing the genetic codes needed to make antigens from scratch. The body’s own cells then secrete the antigens and the immune system responds to them in the same way as it would react to a conventional vaccine. Another method under development
in 2014 is the use of vector vaccines. In these cases, harmless viruses or bacteria are used as carriers to transport the DNA encoding the antigens of disease-causing microorganisms. These vectors mimic
an infection, but because they are harmless, they do not cause disease. The antigens are still displayed, however, allowing the immune system to learn to recognise them.
Many vaccines contain additional ingredients. Aluminium-based adjuvants are often added to enhance the immune response, thereby making vaccines more effective. They work by helping the immune system to
see the antigens in the vaccine. This reduces the amount of microorganism or antigen needed, and may reduce the number of repeat vaccinations required, thereby reducing the cost. Vaccines also often contain stabilisers or preservatives to prolong
their lifespan and improve their storage.
As with other medicines, vaccines must be tested in clinical trials to demonstrate that they are effective and to assess side effects. This process generally takes several years, but can be even longer for vaccines as they are generally not used to treat patients, but are tested in healthy people who are at particular risk of infection.
4. Mode of Administration
Most available vaccines are delivered by injection, either into the fatty tissue just below the skin, or into muscle. A number of oral vaccines have also been developed. These are easier to administer, but because they pass through the digestive tract this method is not suitable for all vaccine types, because antigens may be altered by the body’s digestive system. The influenza vaccine for children is, as of early 2014, currently the only vaccine that can be administered through the nasal passage. It is given in the form of a spray that is directed into the nose and inhaled through normal breathing. Jet-injectors, which shoot a high-pressure stream of liquid medication through the skin to underlying tissues, were first used for medical applications in the 1940s. These were further developed in the late 20th and early 21st centuries and are still being optimised for use with vaccines now.5. Example - The measles, mumps and rubella (MMR) vaccine
The MMR vaccine is a combination vaccine against measles, mumps and rubella. Measles and mumps are highly contagious diseases with life-threatening complications, such as inflammation of the brain (encephalitis) and infection of the membranes that surround the brain and spinal cord (meningitis). Rubella infection results in a rash and cold-like symptoms. It is usually a mild disease, but in pregnant women it can harm the unborn baby, causing birth defects or even miscarriage. The MMR vaccine is given as a single injection into the muscle of the thigh or upper arm. It is a live-attenuated vaccine, so the vaccine contains weakened forms of each of the three disease-causing microorganisms. It is first given to babies at 12–13 months of age, with a booster due when children are between 3 and 5 years old. This provides long-term protection against the three diseases. The most common side effects are the development of very mild forms of measles and mumps that typically last for just a couple of days.The MMR vaccine was introduced in the late 1980s and has radically reduced the number of cases of measles, mumps and rubella. Thanks to the vaccination programme, measles became extremely rare in Europe during the 1990s; however, a research paper published in 1998 suggesting a link between the MMR vaccine and autism resulted in vaccination rates falling in Europe. This research has since been entirely discredited because of false and unreliable results, but the fall in vaccination rates has led to several measles outbreaks across Europe. Catch-up vaccination programmes are now in place to increase the proportion of the population protected against these diseases.
6. Example - Cancer vaccines
The first preventive cancer vaccines, such as human papillomavirus vaccine, act against the human papilloma virus (HPV) which can cause cervical cancer. Human papilloma virus (HPV) is a family of viruses. Some types cause cold sores, warts and verrucae, while others can lead to cancer. Two types of HPV, types 16 and 18, are responsible for over 70% of cases of cervical cancer. Three doses of the vaccine are administered by intramuscular injection over a 12-month period. The most common side effects are pain, swelling and redness at the injection site and headache. In clinical trials in young women with no evidence of previous infection, HPV vaccines were over 99% effective at preventing pre-cancerous lesions associated with HPV types 16 or 18.
Sipuleucel-T is the first approved therapeutic cancer vaccine. It is used in men with metastatic castration-resistant (hormone-refractory) prostate cancer with either no or minimal symptoms. Most prostate cancers express a protein called prostatic acid phosphatase (PAP) on the surface of their cells. The immune system does not normally recognize PAP as foreign and so does not attack and destroy the cancer cells. The process for treating patients with the sipuleucel-T vaccine involves firstly taking blood from a patient and extracting some of the blood cells in the laboratory (Figure 2). These blood cells are then grown in the laboratory alongside a special version of PAP that has been modified by being attached to another molecule called granulocyte-macrophage colony-stimulating factor (GM-CSF). The PAP antigen and GM-CSF are taken up by the immune cells within the blood cells being grown in the laboratory, at which point the GM-CSF helps to activate them. The activated immune cells are then infused back into the patient. These cells interact with other components of the immune system and instruct them to identify the prostate cancer antigen (PAP) as foreign, thereby vaccinating the patient against the prostate cancer. The immune system therefore attacks and destroys the cancer.
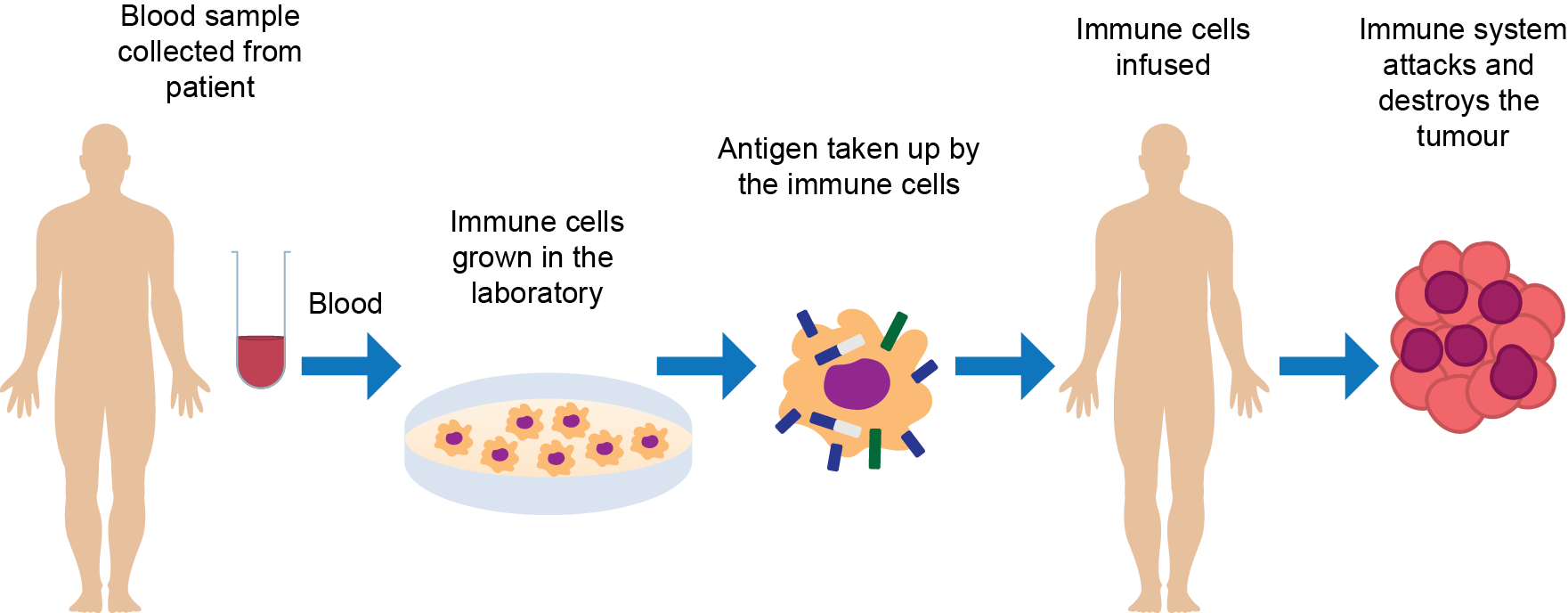
Figure 2. Sipuleucel-T vaccine. Immune cells are extracted from a patient’s blood and grown in the laboratory with an altered form of the prostate cancer antigen, PAP, that has been attached to a GM-CSF molecule. The immune cells are activated by the PAP-G-CSF, which become primed to identify PAP as a ‘foreign’ antigen. These cells are then infused back into the patient, where they instruct other immune cells to attack and destroy the tumour . (GM-CSF, granulocyte-macrophage colony-stimulating factor; PAP, prostatic acid phosphatase)
7. Further Reading
- Vaccines Europe. About vaccines. Available from: About vaccines - Vaccines Europe (Accessed 2 March 2022).
- Vaccines Today site: The target audience is the general public and others in Europe with an interest in immunisation: VaccinesToday Information on Covid 19 vaccines can be accessed directly at: COVID-19 vaccines - VaccinesToday
- World Health Organization Vaccines and immunization (who.int) (accessed 2 March 2022)
8. References
- The Jenner Institute, 2013. Edward Jenner. Available from: Home — The Jenner Institute
- National Health Service Choices, 2013. MMR vaccine side effects. Available from: MMR (measles, mumps and rubella) vaccine - NHS
- Department of Health (UK), 2012. The ‘Green Book’ chapter on human papillomavirus (HPV ). Available from: Human papillomavirus (HPV): the green book, chapter 18a - GOV.UK
- European Medicines Agency, 2013. Provenge. Available from: Provenge | European Medicines Agency (europa.eu)